Occurrence and Environmentally Friendly Control of Rice Stem Borer, Chilo suppressalis in Amur Silver Grass (Mischanthus sacchariflorus) Plantation
Abstract
Insect pests and environmentally friendly control agents were investigated from artificially made Amur silver grass (Miscanthus sacchariflorus) plantations in Iksan (Jeonbuk province), Hwasun (Jeonnam province), and Sancheong (Gyeongnam province). In addition, environmentally friendly control of rice stem borer (RSB), Chilo suppressalis was made with entomopathogens isolated from the given plantations and commercial products in laboratory and field. RSB was confirmed as a major pest in the Amur silver grass which had received attention as a biofuel feedstock in recent. In the light of brief ecology of RSB, overwintering larval density was the highest in Hwasun plantation as 3.7/m2, followed by Sancheong plantation as 0.1/m2. Although overwintering larvae were not found from Iksan plantation, there were lots of damage traces in the plants. Emergence time of 1st generation of RSB was early May in all plantations. Environmentally friendly control agents used in the experiments were entomopathogenic nematodes (Korean indigenous Steinernema carpocapsae Pocheon strain, S. carpocapsae Iksan strain, S. monticolum Hwasun strain, and Heterorhabditis megidis Hwasun strain), fungus (Beauveria bassiana isolated from Hwasun), and commercial Bacillus thuringiensis var. kurstaki. In the environmentally friendly control of RSB, S. carpocapsae Iksan strain, B. bassiana, and H. megidis Hwasun strain isolated from plantations were more effective than S. carpocapsae Pocheon strain in the field as 80.6%, 60.2%, and 58.3% efficacies, respectively. However, S. carpocapsae Pocheon strain was more effective in the laboratory, that is, 49.6% mortality was shown in the S. carpocapsae Pocheon strain, followed by 24.8% in the H. megidis Hwasun strain, 20.4% in the S. carpocapsae Iksan strain, 12.0% in the S. monticolum Hwasun strain, and 6.4% in the B. bassiana Hwasun isolate. Because application of chemical insecticides is not desirable to control insect pests in the Amur silver grass plantations around or close to river, environmentally friendly control agents, especially entomopathogens isolated from plantations are positively recommended for the control of RSB in the Amur silver grass plantations.
초록
최근 바이오 연료의 공급원으로 주목 받고 있는 거대억새(Miscanthus sacchariflorus)의 안정된 생산을 위하여 전북 익산과 전남 화순, 그리고 경남 산청에 인위로 조성한 거대억새 재배지에서 해충의 종류와 친환경적 방제인자를 조사하였다. 그 결과, 이화명나방(Chilo suppressalis)이 주요 해충으로 확인되었다. 그리고 월동 유충의 밀도는 화순에서 3.7마리/m2로 가장 높았으며 다음으로 산청에서 0.1마리/m2였다. 익산에서는 월동 유충이 발견되지 않았으나 억새에서는 피해흔적을 확인할 수 있었다. 1세대의 우화시기는 모든 재배지에서 5월 초순이었다. 한편, 친환경적 방제를 위해 사용한 인자들은 거대억새 재배지에서 분리한 곤충병원미생물을 포함한 곤충병원성 선충(Steinernema carpocapsae 포천 계통, S. monticolum 익산 계통, H. megidis 화순 계통), 곰팡이(Beauveria bassiana 화순분리균) 및 시판 Bacillus thuringiensis var. kurstaki이었다. 재배지에서는 분리된 S. carpocapsae 익산 계통과 화순 재배지에서 분리한 B. bassiana 및 H. megidis 화순 계통의 효과가 80.6%, 60.2%, 58.3%로 S. carpocapsae 포천 계통보다 좋았다. 그러나 실내에서는 S. carpocapsae 포천 계통이 49.6%, H. megidis 화순 계통이 24.8%, S. carpocapsae 익산 계통이 20.4%, S. monticolum 화순 계통이 12.0%, B. bassiana 화순 분리균이 6.4%의 효과를 보였다. 강 주변이나 강 가까운 거대억새재배지에서는 해충의 방제를 위하여 농약을 사용할 수 없기 때문에 널리 사용하고 있는 친환경방제인자, 특히 재배지에서 분리한 곤충병원성 방제인자를 이화명나방의 방제에 적극적으로 활용할 것을 권장한다.
Keywords:
Amur silver grass, environmentally friendly control, Beauveria bassiana, entomopathogenic nematodes, Steinerma, Heterorhabditis키워드:
거대억새, 친환경방제, 곤충병원성선충Introduction
The rice stem borer (RSB), Chilo suppressalis (Walker) is widespread species and serious pest of rice. Apart from the major food plant, rice, RSB larvae also feed on wide range of plants such as Gigantochloa verticellata, Echinochloa crusgalli cruspavonis, Echinochloa stagnina, Eleusine indica, Panicum sp., Paspalum conjugatum, Amaranthus sp., Phragmites australis, Raphanus raphanistrum, Sclerostachya fusca, Sorghum sp., Typha latifolia, Xanthium strumarium, and Zizania aquatica (https://en.wikipedia.org/wiki/Chilosuppressalis). Because RSB has low temperature tolerance, it also adapts harsh winter of Korea. In Korea, it has been the most serious pest of rice until late 1960s (Lee and Park, 1991), but change of rice varieties and cultural practices resulted in low population or non-occurrence of RSB on rice for the last several decades (Ryoo and Lee, 1985). Consequently, RSB has been neglected in the rice farming in recent. However, it is different story in the artificially made Amur silver grass (Miscanthus sacchariflorus) plantations because RSB became major pest of Amur silver grasses in the plantations, especially in Hwasun (Ahn, personal communication). Amur silver grass has received attention as potential biofuels feedstock because it consists of low lignin (about 20-24%) and high polysaccharide contents (nearly 65%), indicating that their great proportion of biomass could be used as bioenergy (Kim et al., 2012). In recent, the Amur silver grass plantations were artificially made in some areas around four major rivers of Korea including Iksan plantation (Jeonbuk province) covering an area of 60,000 m2, Hwasun plantation (Jeonnam province) covering an area of 7,500 m2, and Sancheong plantation (Gyeongnam province) covering an area of 6,600 m2. Because plantations are located around or close to river, chemical insecticides are not desirable to control insect pests of silver grasses in the plantations.
If there are any effective environmentally friendly control agents available, these agents can be strongly required for the control of insect pests of Amur silver grass. Thus, insect pests and environmentally friendly control agents were investigated from these plantations.
In the course of investigation of insect pests from the given plantations, RSB was confirmed as the most serious pest out of detected insect pests. Although RSB is a major pest in the Amur silver grass plantations, any study on RSB was not made from Amur silver grass yet. Therefore, brief ecological investigation of RSB was made from the given Amur silver grass plantations.
In addition, environmentally friendly control was attempted in laboratory and plantation. Out of environmentally friendly control agents, entomopathogenic nematodes are promising against RSB (Choo et al., 1991). Entomopathogenic fungi are also promising agents and being widely used against important pests (Vanesa, 2011). Thus, Korean indigenous entomopathogenic nematodes and entomopathogenic fungus were used as environmentally friendly control agents against RSB.
In fact, entomopathogenic nematodes in the genera Steinernema (Steinernematidae) and Heterorhabditis (Heterorhabditidae) are obligate important natural mortality factors of insects having useful attributes such as wide host range, easy application in the field, safety, and active foraging of hosts in the ecosystem (Susurluk, 2011). Another potential agent group, entomopathogenic fungi are usual natural enemies of arthropods worldwide, occupying virtually every niche in which arthropods are also found (Vanesa, 2011). Therefore, these environmentally friendly control agents including commercial Bacillus thuringiensis (Bt) were used for the control of RSB in the laboratory and Amur silver grass field.
Materials and Methods
Studied site
Insect pests and environmentally friendly control agents, especially microbial pathogens were investigated from three artificially made Amur silver grass plantations where were located around or close to river in Iksan, Jeonbuk province (Iksan plantation built in 2011: 36o8'44.22''N, 126o56'22.26''E; 60,000 m2 in size), Hwasun, Jeonnam province (Hwasun plantation built in 2010: 34o59'33.66''N, 127o5'15.05''E; 7,500 m2 in size), and Sancheong, Gyeongnam province (Sancheong plantation built in 2010: 35o16'3.72''N, 127o532'34.98''E; 6,623 m2 in size). The Geumgang River was flowing down to the northwest and rice was cultivating to the southeast of Iksan plantation. The Eoenam stream was flowing down to the west of Hwasun plantation and rice was cultivating around plantation. The chestnut orchard was 15 m apart from Hwasun plantation to the east. The Deogcheon River was flowing down to the east of Sancheong plantation. Rice, watermelon, and melon were cultivating in the summer, but strawberry was cultivating from the fall in the polyethylene film houses to the south of plantation. The RSB studied area from each plantation was 2,000 m2 out of 60,000 m2 in Iksan plantation, 2,700 m2 out of 7,500 m2 in Hwasun plantation, and 3,500 m2 out of 6,600 m2 in Sancheong plantation, respectively. Investigation was made from April to November from 2013 to 2014.
Investigation of overwintering RSB larvae
Overwintering RSB larvae were investigated twice a month in February and April from the given plantations. Each plantation was divided into nine rectangle sampling sets which are of a size. In each sampling set, one 1 m2 area was randomly selected for subsampling site of RSB larvae. The stubble of silver grass was pulled up by the roots from each subsampling site and transported to the laboratory. Then, roots were carefully washed, cut, and split to take out RSB larvae. When RSB larvae were found, the number of larvae per plant was recorded. Sampling dates were 5 February and 3 April in Iksan plantation, 7 February and 4 April in Hwasun plantation, and 6 February and 2 April in Sancheong plantation, respectively.
Investigation of emergence time of RSB
Five emergence traps sizing 90 cm × 90 cm × 90 cm were installed in each plantation in Iksan, Hwasun, and Sancheong on 2, 4, and 9 April in 2013, respectively. The emergence trap was covered with insect net and fixed with soil around bottom of each trap. Then, the yellow sticky trap was hung in the center of emergence trap. The emergence of RSB was checked three times every two weeks from mid-April. In 2014, observation was made only in Hwasun plantation on 7, 16, and 28 May, where received severe damage by RSB.
Damage of Amur silver grass by RSB
Damage rate of Amur silver grass by RSB larvae was investigated from the given plantations in Iksan, Hwasun, and Sancheong from 2013 to 2014. In 2013, Iksan and Sancheong plantations where overwintering RSB population was low were divided into 33 sections. On the other hand, Hwasun plantation where high population of overwintering RSB was recorded was divided into 48 sections. In 2014, each plantation was divided into 30 sections. One hundred Amur silver grasses were randomly selected from each section and checked damage by RSB symptom. When silver grasses were damaged by RSB larvae, stems of silver grasses were easily distinguished by entrance hole and frasses on stems and fallen down of plant thereby. The first investigation was made from late June to early July in the first generation and the second was made in the middle of September in the second generation.
Damage position of RSB on Amur silver grass stem
Damage position of RSB was investigated from silver grass stems. Two hundred forty-two damaged silver grasses were randomly collected from Hwasun plantation. The plant was cut from the bottom with pruning shears and transported to the laboratory. The stems were observed at intervals of 20 cm and damage hole was checked. Observation was made on 12 November, 2013.
RSB larvae for environmentally friendly control experiment in the laboratory
RSB larvae were collected from RSB infected silver grasses and healthy larvae were used. Enough infected grass stems were cut and brought to the laboratory from the plantations. The stems were left for one week to obtain healthy larvae. Larvae, that is, were taken out from stems with frasses and healthy larvae were used.
Environmentally friendly control agents
Korean indigenous entomopathogenic nematodes, S. carpocapsae Pocheon strain, S. carpocapsae Iksan strain, S. monticolum Hwasun strain in the Steinernematidae and Heterorhabditis megidis Hwasun strain in the Heterorhabditidae, entomopathogenic fungus, Beauveria bassiana isolated from Hwasun, and commercial B. thuringiensis var. kurstaki were used. Iksan and Hwasun strains of entomopathogenuic nematodes and fungus were isolated from studied plantations.
Nematodes
The entomopathogenic nematodes were isolates from soil using the Galleria mellonela trapping method (Bedding and Akhurst, 1975). Each of the Korean isolates was reared in G. mellonella larvae using the method of Dutky et al. (1964). The infective juveniles were collected in White traps, harvested, and stored at 10oC for no more 3 weeks before they were used (Kaya and Stock, 1997).
Fungus
B. bassiana was isolated from soil of Hwasun plantation. Culturing and preparation of fungus for the experiment were followed by Choo et al’s method ((Choo et al., 2002). That is, ten G. mellonella larvae were exposed to the fungal laden cadaver from trapped container. After 12 h, the cadaver on a moist piece of filter paper in a Petri dish was removed and the Petri dish was placed in a plastic bag at 25oC. Ten days after exposure, the G. mellonella cadavers in the Petri dish were stored in a refrigerator at 5oC. To obtain conidia for the experiments, G. mellonella cadavers were removed from the refrigerator and 10 G. mellonella were exposed to each cadaver as described above. Eight days later, the dead Galleria larvae were surface sterilized in 1% NaOCl and 75% EtOH for 1 min each followed by three rinses in distilled water. The surface-sterilized cadavers were placed on 1% sterilized water agar for 7 days to allow sporulation. The conidia were harvested aseptically and placed in sterilized distilled water containing 0.2% of the wetting agent Tween 20 (Duksan Pharmaceutical Co. Yongin, Korea). This conidial suspension was used for mass production of the fungus on potato dextrose agar (PDA) for two weeks. The concentration of conidia was determined by using a dilution series on PDA. The conidial suspension was diluted by a 10-fold dilution series and 100 μl of the suspension was plated on the agar. Cultures were incubated for 3 to 4 days at 25oC and the number of colonies was counted at the dilution providing between 50 and 200 colony forming units (CFUs)/dish was calculated (Choo et al., 2002). There were three replicates per dilution
Bt
Bt used in the experiment was commercial B. thuringiensis var. kurstaki (serotype IIIa, IIIb, 16 BIU/kg). The wettable powder of Bt (Brand name: Scorpion, Haneol Science, Seongnam, Gyeonggi province, Korea) was purchased from agricultural chemicals market and used as recommended dosage.
Laboratory test
The silver grass stems were cut by 20 cm centering around entrance hole and put them into wet flower-pack (230 × 110 × 70 mm). Each flower-pack contained 10 stems and received 3 ml of 1,000 infective juveniles (IJs) of given nematodes and 1 × 108 conidia of fungus by hand sprayer. B. thuringiensis var. kurstaki was treated as recommended concentration, 1,000 times. 3 ml of chlorantraniliprole which was registered for RSB control was also treated by 2,000 times to compare efficacy with entomopathogenic nematodes, fungus, and bacterium. The control received only 3 ml of water. Treated stems were kept at 25 ± 2oC for one week. One week later, stems were carefully taken out, cut, and split. Then, RSB larvae were checked. There were three replicates in all agents.
Concentration test of S. carpocapsae Pocheon strain
S. carpocapsae Pocheon strain which was the most effective in the laboratory experiment was tested again to make a decision of optimum concentration. The silver grass stems were prepared as described above and received 3 ml of 0, 250, 500, 1,000, 2,000, and 4,000 IJs. The treated containers and RSB larvae were kept and checked as above. There were three replicates per concentration.
Field test
In 2014, 2 m × 2 m plots with 1 m buffers between the plots were established in the Hwasun plantation, where was more RSB infected. The following treatments were applied; (1) S. carpocapsae Pocheon strain 4 × 105 IJs (=1 × 109 IJs/ha), (2) S. carpocapsae Iksan strain 4 × 105 IJs (=1 × 109 IJs/ha), (3) H. megidis Hwasun strain 4 × 105 IJs (=1 × 109 IJs/ha), (4) Beauveria bassiana Hwasun isolate 1 × 1012CFU (=2.5 × 1015CFU/ha), (5) B. thuringiensis var. kurstaki (Brand name, Scorpion K, Haneol Science, Korea) 3 g/3 l (=30 × 106 cfu/kg), (6) carbofuran 3 g/m2, and (7) Control. The applications were made between 16:00 and 17:30 h. The weather was sunny and ambient temperature 20.1oC. The average soil moisture was 20.2%. Two and three days later after application, it rained 14.5 mm and 2.0 mm, respectively. After treatment, 3 liters of water was applied per plot. The plots were covered with mosquito net and yellow sticky trap was installed in the center of the net. The plots were set up in a completely randomized block design with three replicates. One month later, emerged adults were counted three times every two weeks.
Statistical analysis
Damage height of RSB, pathogenicity of entomopathogens against RSB larvae, and effect of concentrations of S. carpocapsae on pathogenicity were subjected to analysis of variance (ANOVA) and the differences among treatment means were compared using a Tukey’s studentized range test (P/0.05). All statistical analyses were conducted using SAS (SAS Institute 1999).
Results
Density of overwintering RSB larvae
Infection rates of Amur silver grass by RSB were 21.6% and 16.1% in Hwasun plantation on 7 February and 4 April, respectively (Table 1). Larval density was from one to twelve averaging 3.7 from a plot. However, none was found from the plot in Iksan plantation and one from the plot in Sancheong plantation on the surveying date of 2 April.
Emergence time of RSB
RSB started emergence from early May in Sancheong and Hwasun, but from mid-May in Iksan (Table 2). The number of emerged adults was low in Sancheong and Iksan plantations where larval densities were low while more adults were recorded in Hwasun plantation where larval density was higher. No emergence was observed after early June. More emerged adults were recorded in 2014 than 2013.
Damage rate of RSB
In 2013, damage rate of Amur silver grass was higher in the first generation of RSB than the second generation, that is, damage rate of Amur silver grass by RSB from Hwasun plantation was 27.6% on 28 June while 11.9% on 13 September, respectively (Table 3). However, more damaged stems were observed in the second generation of RSB in Iksan and Sancheong plantations. Damage was not observed on 13 June with the naked eyes, but 25% of silver grasses fallen down by RSB infection was observed (Observation data). On 13 September, damage was not found from the fallen silver grasses, but found from standing silver grasses. Entrance holes in the second generation were higher than ones in the first generation of RSB (Observation data). Less than 1% damage was observed from Iksan and Sancheong plantations. In 2014, damage rate was 26.7% and 23.3% in Hwasun plantation in the first and second generation, respectively. However, damage rate of the first and second generations was 9.1% and 7.7% in Iksan plantation and 1.1% and 2.7% in Sancheong plantation, respectively.
Damage position of RSB on the stem of Amur silver grass
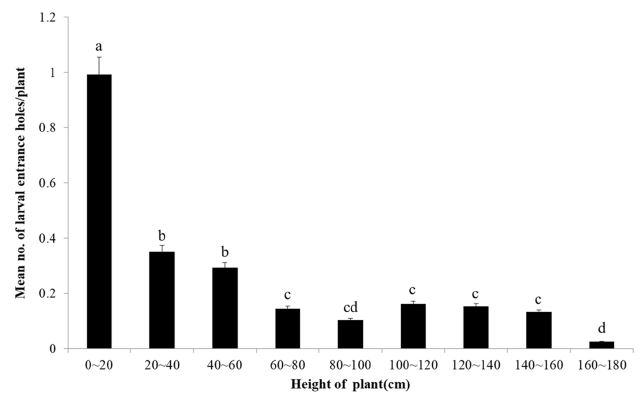
One to six holes were found from the stem of silver grasses and most holes were distributed below 1 m. Damage position of RSB was different (F = 71.91; df = 8, P = <0.0001). Most of damage position was 0 to 20 cm and more was found lower part than upper part (Fig. 1), that is, 4 larvae were found between 0 – 20 cm while 1 was found between 40 – 60 cm. Fallen grasses were observed from July.
Pathogenicity of environmentally friendly control agents against RSB larvae in the laboratory
Pathogenicity of entomopathogenic agents were different depending on entomopathogens, that is, 49.6% mortality was shown in the S. carpocapsae Pocheon strain, 24.8% in the H. megidis Hwasun strain, 20.4% in the S. carpocapsae Iksan strain, 12.0% in the S. monticolum Hwasun strain, 6.4% in the B. bassiana Hwasun isolate, 3.7% in the commercial B. thuringiensis var. kurstaki, and 10% in the chemical insecticide, respectively (Fig. 2).
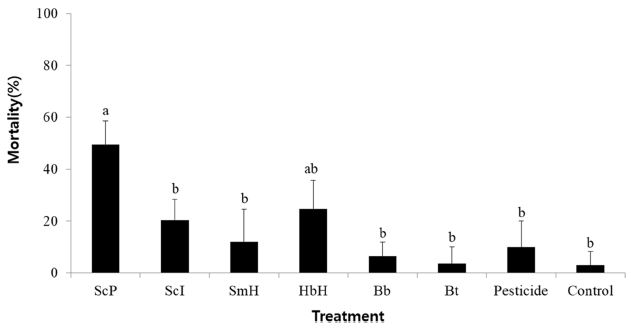
Efficacy of environmentally friendly control agents against RSB in the laboratory. ScP: Steinernema carpocapsae Pochen strain, ScI: S. carpocapsae Iksan strain, SmH: S. monticolum Hwasun strain, HmH: H. megidis Hwasun strain, Bb: Beauveria bassiana Hwasun isolate, Bt: commercial Bacillus thuringiensis var. kurstaki, Pesticide: chlorantraniliprole 4% WP. Different letters above standard deviation bars indicate significant differences among means at Type 1 error = 0.05 (Tukey’s HSD test).
Pathogenicity of S. carpocapsae Pocheon strain against RSB depending on concentration
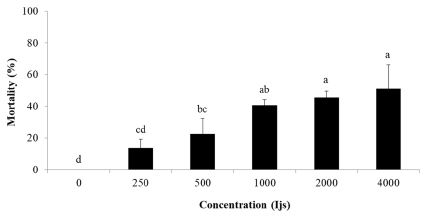
S. carpocapsae Pocheon strain showing the highest pathogenicity was evaluated again against RSB larvae depending on concentrations. The mortality was higher with increasing concentrations, but not much different at the rate of >1,000 IJs (Fig. 3). The mortality was 13.7%, 22.5%, 40.6%, 45.4%, and 51.0% at the rates of 250, 500, 1,000, 2,000, and 4,000 IJs, respectively. Therefore, optimum concentration of S. carpocapsae Pocheon strain was 1,000 IJs/ml.
Field efficacy of environmentally friendly control agents against overwintering RSB larvae in the field
Not like laboratory test, S. carpocapsae Iksan strain showed the highest efficacy in the field based on the emerged number of adults (Fig. 4). The number of adults emerged was 6 at the plot of S. carpocapsae Iksan strain. However, there were 22 adults at the plot of S. carpocapsae Pocheon strain which was the most effective agent in the laboratory test. On the contrary, 13 and 12 adults were recorded at the plots of H. megidis Hwasun strain and B. bassiana Hwasun isolate, respectively (Table 4). The rates of control were 80% at the treatment of S. carpocapsae Iksan, 61.2%, and 58.3% at the treatments of B. bassiana Hwasun isolate and H. megidis Hwasun strain, respectively (Fig. 4). Bt and chemical insecticide, carbofuran were less effective than entomopathogenic nematodes and fungus isolated from the studied plantations.
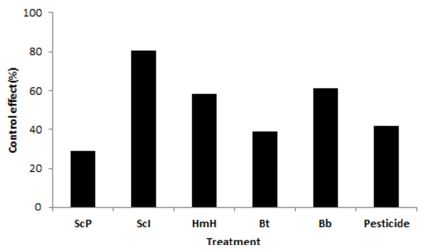
Field efficacy of environmentally friendly control agents against RSB. ScP: Steinernema carpocapsae Pochen strain, ScI: S. carpocapsae Iksan strain, HmH: H. megidis Hwasun strain, Bb: Beauveria bassiana Hwasun isolate, Bt: commercial Bacillus thuringiensis var. kurstaki, Pesticide: carbofuran 3% GR.
Discussion
Miscanthus sacchariflorus is a perennial fast growing grass. In recent, this grass has received attention not only as bioenergy plant, but also as ornamental plant, vegetative filtering plant for cleaning water, and shelters for wild birds. Thus, Amur silver grass has been artificially planted around major rivers and many idle lands according to purposes. Especially, Amur silver grass plantations were made around or close to four major rivers in Korea. Agricultural chemicals, therefore, are not recommended for the control of pests of Amur silver grass when insect pests become an issue. Because insect pests on silver grasses were rarely known in Korea, insect pests and entomopathogens were investigated together from artificially made Amur silver grass plantations. In the course of investigation of insect pests for the sound production of Amur silver grass as bioenergy feedstock, RSB was confirmed as a major pest in the Amur silver grass plantations although population was different depending on studied plantations. Hwasun plantation was more damaged than Iksan and Sancheong plantations. More RSB occurrence in Hwasun plantation might be resulted from rice cultivation close to plantation and outbreak of RSB in the preceding year. Artificial and natural barriers, big and high bank or river was in front of Iksan and Sancheong plantations. These barriers could contribute to interruption of insect pest immigration from neighboring fields to the plantations. However, silver grass can be damaged even at low density because one larva can attack several stems in the field (Kim et al., 2012). This could explain silver grass damage in Iksan plantation even at low density. That is, damage traces of RSB were high in the fields although low density was recorded from Iksan and Sancheong plantations.
It is known that overwintering RSB larvae in the inside of stem or rice stubbles move to cutting part of stem from mid-April and pupate, and then start to emerge from early May (Kim et al., 2012). Although host is different, emergence time of RSB was the same as recorded on rice in the literatures. RSB prefers lower part of stem to upper part, that is, lower part of grass stem below 20 cm was more damaged by RSB than upper part of stem. RSB damage was rarely found from the upper part of grass stem in our study.
Damage of lower part of stems leads to severe results falling down of whole plant in the end. Thus, our observation came to a conclusion that RSB was the most serious insect pest of Amur silver grass and could damage silver grasses even at low density.
Because insect pests occurring in Amur silver grass plantations close to rivers are not desirable to be controlled by chemical insecticides, alternative tactics have to be adapted in the silver grass plantation. Entomopathogenic nematodes, fungi, and Bt are potential alternatives. Thus, these entomopathogens were evaluated against RSB in the laboratory and field. Promising environmentally friendly control agents were also detected simultaneously with insect pests from studied Amur silver grass plantations. Fortunately, some potential entomopathogenic agents were isolated from plantations such as S. carpocapsae, S. monticolum, H. megidis, and B. bassiana. In fact, entomopathogenic nematodes, S. carpocapsae and H. bacteriophora are effective against rice stem borer when sprayed on rice stems. H. bacteriophora was more effective than S. carpocapsae in the laboratory test (Choo et al., 1991). Caterpillars are generally susceptible to Steinernema nematodes. It was also effective against RSB in our laboratory study. However, efficacies in the field study were different from those in laboratory study. Sometimes environmentally friendly control agents co-existed with pests can be more effective than introduced ones. S. carpocapsae Iksan and H. megidis Hwasun strains, and B. bassiana isolated from Hawsun plantation were actually more effective showing 80% and 60% in the field, respectively, although S. carpocapsae Pocheon strain was more effective in the laboratory. The efficacies of entomopathogenic nematodes and fungus from our study gave positive hope of using these entomopathogens against RSB in Amur silver grass plantations. There are two types of foraging behaviors in entomopathogenic nematodes, cruise (widely foraging) and ambush (sit-and-wait) (see Choo et al. 2011). Our results showed that both types were effective against RSB larvae although the cruiser has superior host finding ability to the ambusher (Choo et al. 1989). S. carpocapsae is an ambush type while H. megidis is a cruise type.
Besides effectiveness of entomopathogenic nematodes against insect pests, entomopathogenic fungi such as B. bassiana, B. brongniartii, and Metarhizium anisopliae are also promising and receiving an interest for the control of insect pests. For example, B. brongniartii propagated on rice bran was effective against Exomala orientalis in golf courses (Choo et al., 2002). In addition, it lasted long time at the applied field and easily propagated on media. Efficacy of B. bassiana was also recorded from many important insect pests from many countries (see Vanesa, 2011). Results of our study and other workers encouraged to make use of entomopathogenic fungi for the environmentally friendly control of RSB on Amur silver grass. Ineffectiveness of chemical insecticides might be resulted from characteristic of silver grass. Slippery surface of silver grass and entrance hole of RSB made unfavorable for insecticides to reach RSB.
Moreover, young silver grasses are suitable for the use of entomopathogens and that entomopathogenic nematodes and fungi are easy to apply in the 1st generation of RSB.
Thus, these promising environmentally friendly control agents are positively recommended to control insect pests including RSB for the sound production of Amur silver grasses in the plantations.
Acknowledgments
This research was supported by the RDA Agenda projects (PJ0092422013).
References
- Bedding, R. A., and R. J. Akhurst, (1975), A simple technique for the detection of insect parasitic nematodes in soil, Nematologica, 21(1), p109-110.
- Choo, H. Y., H. K. Kaya, T. M. Burlando, and R. Gaugler, (1989), Entomopathogenic nematodes: Host-finding ability in the presence of plant roots, Environmental Entomology, 18(6), p1136-1140.
- Choo, H. Y., H. K. Kaya, J. B. Kim, and Y. D. Park, (1991), Evaluation of entomopathogenic nematodes, Stainernema carpocapsae (Steinernematidae) and Heterorhabditis bacteriophora (Heterorhabditidae) against rice stem borer, Chilo suppressalis (Walker) (Lepidoptera: Pyralidae), Korean J. Appl. Entomol., 30(1), p50-53.
- Choo, H. Y., H. K. Kaya, J. Huh, D. W. Lee, H. H. Kim, S. M. Lee, and Y. M. Choo, (2002), Entomopathogenic nematodes (Steinernema spp. and Heterorhabditis bacteriophora) and fungus Beauveria brongniartii for biological control of the white grubs, Ectinohoplia rufipes and Exomala orientalis, in Korean golf courses, BioControl., 47(2), p177-192.
- Dutky, S. R., J. V. Thompson, and G. E. Cantwell, (1964), A technique for the mass propagation of the DD-136 nematode, J. Insect Pathol., 6(4), p417-422.
- https://en.wikipedia.org/wiki/Chilo, suppressalis
- Kaya, H. K., and S. P. Stock, (1997), Technique in insect pathology, In: Manual of techniques in insect pathology, L. A. Lacey (ed.), Academic Press, SanDiego, CA, USA, p281-324.
- Kim, S. J., M. Y. Kim, S. J. Jeong, M. S. Jang, and I. M. Chung, (2012), Analysis of the biomass content of various Miscanthus genotypes for biofuel production in Korea, Industrial Crops and Products, 38, p46-49.
- Kim, G. H., D. S. Kim, C. G. Park, S. W. Cho, Y. N. Youn, and K.Y. Lee, (2012), Principles and applications in insect pest management, Hywaengmoonsa, Seoul, Korea, p478.
- Lee, S. C., and H. J. Park, (1991), Changes in the occurrence pattern of the striped rice borer, Chilo suppressalis Walker, in Korea, Korean J. Appl. Entomol., 30(4), p249-257.
- Ryoo, M. I., and M. H. Lee, (1985), Characteristics of the aggregation pattern of the striped rice stem borer (Chilo suppressalis (Walker)) during the larval stage, Korean J. Appl. Entomol., 24(1), p1-6.
- SAS Institute, (1999), STATVIEW, SAS Institute, Inc., Cary, NC.
- Susurluk, I. A., (2011), Isolation, preservation, characterization and identification of entomopathogenic nematodes, In: Microbial insecticides, principles and applications, Borgio, J.F., Sahayaraj, K., and Susurluk, I. A. (eds), Nova Science Publishers, Inc., New York, USA, p193-210.
- Vanesa, T. A., (2011), Biological control potential of entomopathogenic fungi, In: Microbial insecticides, principles and applications, Borgio, J.F., Sahayaraj, K., and Susurluk, I. A. (eds), Nova Science Publishers, Inc., New York, USA, p29-46.