Evaluation of fungicidal control of garlic leaf blight using mobile spraying equipment
 ; Wooman An1 ; Sungyu Choi1
; Wooman An1 ; Sungyu Choi1 ; Hyeongju Choi1
; Hyeongju Choi1 ; Jae-Ha Choi1
; Jae-Ha Choi1 ; Soyoon Park1
; Soyoon Park1 ; Hyeong-Rok Jang1
; Hyeong-Rok Jang1 ; Chang-Gi Back2 ; Se-Woon Hong3
; Chang-Gi Back2 ; Se-Woon Hong3 ; Hyunkyu Sang1, *
; Hyunkyu Sang1, *
Abstract
Unmanned aerial vehicle (UAV) - mediated pesticide spraying has established an efficient control system for various pathogens. However, concentrations of fungicide authorized for UAV spraying are relatively higher than for backpack spraying; therefore, unwanted dispersion of fungicides can be problematic in plots with various crops in narrow areas, such as in South Korea. In this study, we demonstrated efficient control of garlic leaf blight (Stemphylium vesicarium and Stemphylium eturmiunum) by mobile spraying equipment (MSE), which is an imitation of UAV. To select an effective fungicide for garlic leaf blight, we selected each of the most virulent strains of S. vesicarium and S. eturmiunum from pathogenicity assays for garlic with about 60% of the disease severity index. Then, we selected iprodione which showed highest control activity among 5 tested fungicides in a fungicide efficacy test using MSE and EC50 value estimations. Iprodione solutions at different concentrations were tested to evaluate the efficacy of reduced amounts of fungicide by UAV spraying. The fungicide solution at half concentration of iprodione was sufficient to control garlic leaf blight using MSE. Our results will contribute to developing an efficient fungicide application system via spraying regulated concentrations of fungicides using UAV.
Keywords:
fungicide, garlic, garlic leaf blight, UAVIntroduction
Along with onions and leeks, garlic is one of the beneficial crops for its usage in food and medicinal effects including anticancer and antibacterial properties (Tattelman 2005). In South Korea, garlic is cultivated over an area of 24,700 hectares, producing 320,000 tons annually, primarily in Gyeongsangnam-do and Gyeongsangbuk-do (KOSTAT, 2023). During cultivation of garlic, various pathogens including fungi, virus, bacteria, and nematodes are responsible for leaf disease and yield reduction. Particularly, fungal diseases such as blue mold (Penicillium spp.), black mold (Aspergillus niger), basal rot (Fusarium oxysporum), and rust disease (Puccinia allii) are the most devastating diseases in garlic (Koike et al., 2001; Valdez et al., 2006; Chavda and Brahmbhatt, 2016; Mondani et al., 2020). Stemphylium leaf blight is also a significant problem in the United States and India (Anupriya et al., 2021) and its causal pathogens (Stemphylium vesicarium and Stemphylium eturmiunum) are responsible for yield reduction of garlic and onion. The disease infects primarily the leaves, initially causing small, grayish-white spots that progress to brown spots with black conidia forming on the lesions. First reported in the United States in 1976, leaf blight was not initially a major cause of reduced onion yield. However, since the 2010s, it has been considered a severe pathogen affecting onion production, especially in Canada and New Zealand (Hay et al., 2021). Recently, Stemphylium eturmiunum has been reported as a pathogen causing garlic leaf blight in China and South Korea (Dumin et al., 2022).
Methods of controlling garlic leaf blight include cultural, biological, and chemical. Among the methods, spraying chemicals for control of pathogens is widely used. To spray pesticides, using a recently developed unmanned aerial vehicle (UAV) has been highlighted as a beneficial advance in modern agriculture, capable of spraying a consistent amount of fungicide over a wide range, and studies are being conducted accordingly (Canicattì and Vallone 2024). Fungicides for UAV spraying are authorized at much higher concentrations compared to backpack spraying – about 100-200 times to maximize control efficacy. This leads to increased risk of unwanted dispersion, caused by winds generated from UAV flight. In addition, there are fewer fungicide products authorized for UAV spraying in stemphylium leaf blight of garlic compared to backpack spraying. Therefore, effective and regulated fungicide application using UAV can be accomplished by research of effective fungicides for each disease and application of decreased concentrations of fungicide to reduce the risk of unwanted dispersion. However, very few studies have taken such an approach.
Various fungicides have been used to control garlic leaf blight. Previous studies have shown less, moderate, and high sensitivity of garlic leaf blight to various fungicides: less sensitivity to fungicide groups with EC50 values exceeding 500, 274.1, 65.7, and 144.2 mg L-1 (azoxystrobin, trifloxystrobin, tebuconazole, and carboxin + thiram, respectively); moderate sensitivity to a fungicide with an EC50 value of 39 mg L-1 (difenoconazole); and high sensitivity to fungicides with EC50 values of 4.9, 3, and 1.1 (prochloraz, iprodione, and boscalid + pyraclostrobin, respectively) (Galvez et al., 2016) . In the study by Wang et al., 2021, growth inhibition rates of S. vesicarium by QoI fungicides azoxystrobin, kresoxim-methyl, and pyraclostrobin were less than 17.5% but exceeded 80% for iprodione, difenoconazole, and prochloraz. Additionally, iprodione showed over 60% of conidial germination inhibition activity whereas difenoconazole and prochloraz had 0% activity (Wang et al., 2021).
In this study, we used previously isolated S. vesicarium and S. eturmiunum strains to select the most pathogenic strain from each species and determine the sensitivity of the strains to different classes of fungicides. In addition, efficacy of fungicides to control garlic leaf blight was evaluated by a mobile spraying equipment and dilution effects of the most effective fungicide were tested to determine reduced concentrations of fungicide used for controlling garlic leaf blight. The study provides valuable information for developing a fungicide spray program by UAV.
Materials and Methods
Fungal strains and fungicides
Stemphylium vesicarium strains KACC44528 and KACC44530 causing garlic leaf blight were provided from the Korea Agricultural Culture Collection (KACC, Rural Development Administration, Jeonju, Korea). Stemphylium eturmiunum strains (19-056 and 19-130) and S. vesicarium strains (19-230, 19-238, and 19-241) had previously been isolated from garlic in various regions in Jeollanam-do and Gyeongsangnam-do in South Korea as reported (Back et al., 2022). A total of five fungicides were used in this study – azoxystrobin (Syngenta, Basel, Switzerland), difenoconazole (Syngenta, Basel, Switzerland), iprodione (Syngenta, Basel, Switzerland), florylpicoxamid (Corteva, IN, USA), and penthiopyrad (Syngenta, Basel, Switzerland). Each fungicide at the registered concentration for UAV spraying in South Korea was designated as “1X” concentration; diluted solutions at 66% and 50% concentration of “1X” are designated as “2/3X” and “1/2X”. If a fungicide was not registered for UAV spraying, “1X” concentrations were determined by a specific dilution factor depending on their respective formulation. The “1X” concentrations of active ingredients of each fungicide were determined as follows: 10.85g L-1 for azoxystrobin; 3.125g L-1 for difenoconazole; 26.04g L-1 for iprodione; 6.25g L-1 for florylpicoxamid; and 9.375g L-1 for penthiopyrad.
In vitro fungicide sensitivity assay
For fungicide sensitivity assay of the garlic leaf blight strains, the fungicides (azoxystrobin, difenoconazole, iprodione, florylpicoxamid, and penthiopyrad) were added to PDA (Potato Dextrose Agar) medium at concentrations of 0.01, 0.1, 1, 10, and 100 mg L-1. Salicylhydroxamic acid (SHAM) was added to 100 mg L-1 for azoxystrobin-supplemented PDA. Circular agar plugs, 6 mm in diameter, from colonies of strains grown on PDA were placed on fungicide-supplemented or non-supplemented PDA and cultured at 25°C in darkness. After 8 days of culture, the diameters of the colonies on all the plates were measured by a digital caliper (Mitutoyo, Kawasaki, Japan) and percentage relative mycelial growth (RMG) was calculated. The calculated RMG values on different concentrations of fungicides were subjected to EC50 value estimation using an R studio EC50 estimator package (R core Team, 2021). The experiment was performed with 3 technical replicates and 2 biological replicates.
Pathogenicity assay of garlic leaf blight strains and in planta fungicide control efficacy test
To evaluate the virulence of fungal strains, each strain was cultured in 200 mL of potato dextrose broth (PDB) for 7 days. Each culture saturated with the mycelia of each strain was ground by a commercial blender and sprayed on 3-weeks-old garlic having three to five leaves per plant (n = 6). After 7 days of inoculation, the disease severity index (DSI) was estimated by visual symptoms on the plants: 0 = healthy leaf without lesion; 1-2 = small lesion on tip of leaf; 3-4 = all leaf tips with developed lesions; 5-6 = severe lesion development on all leaves; 7 = plant wilting or death.
For evaluating the control efficacy of fungicides in garlic, fungicides at 1X concentration were sprayed on 2 pots containing six of 3-weeks-old garlics (n = 12) with mobile spraying equipment (MSE). The MSE’s speed was 1 meter per second while spraying fungicide solutions with 250 kPa (2.5 bar) of pressure with the Teejet® XR11002 nozzle connected to a water pump. The nozzle attached to MSE was moved parallel to the ground surface above 1 meter from garlic plants in greenhouse. Mycelial culture was inoculated 3 hours after spraying the fungicides. DSI was evaluated the same as the pathogenicity test of fungal strains. The control efficacy percentage of each treatment was calculated as follows:
Control efficacy (%) = DSI of fungicide treatment / Average DSI of pathogen treatment ×100
To evaluate the control efficacy of different concentrations of fungicide, the fungicide showing the best control efficacy was selected and sprayed on garlic plants (n = 6) at 1X, 2/3X, and 1/2X concentrations. Traveling speed and spraying pressure of MSE were the same as above. Each solution was sprayed 3 hours before inoculation of S. eturmiunum 19-056 and S. vesicarium 19-238 and after 10 and 20 days of inoculation. DSI was evaluated after 30 days of inoculation following the same disease scale of the pathogenicity test. To verify a consistent amount of fungicide was sprayed, water sensitive papers (WSP) were installed on the spraying route of MSE at the same height as the leaf tips of the garlic. Surface coverage of each spraying was calculated by the WSP scanner program (Park et al., 2020) to verify spraying consistent amounts of the fungicide solutions. Statistical difference of means was determined by Fisher’s least significant difference (LSD) test.
Results and Discussion
Estimation of EC50 value of fungal strains
Estimated EC50 values indicated highest mycelial growth inhibition activity of iprodione followed by penthiopyrad, difenoconazole. The highest EC50 values of iprodione, penthiopyrad, and difenoconazole were 1.182(±0.432) mg L-1 of S. eturmiunum 19-056, 1.715(±0.946) and 47.778(±10.161) mg L-1 of S. vesicarium 19-238, respectively. EC50 values of azoxystrobin and florylpicoxamid of some strains were not determined due to less than 50% of mycelial growth inhibition at tested concentrations and designated as “>100” (Table 1). Reduced sensitivity of the recently isolated S. vesicarium and S. eturmiunum strains to azoxystrobin compared to the strains (KACC44528 and KACC44530) isolated in 2006 was observed. The EC50 value of 19-238 strain to difenoconazole was 47.778 mg L-1, which is higher than previously reported in Spain (Galvez et al., 2016). High sensitivity to iprodione and penthiopyrad indicated possible high control efficacy on further in planta fungicide efficacy test. QoI resistance of S. vesicarium has been reported in various countries (Hay et al., 2019; Wang et al., 2021; Spawton et Du Toit, 2024), which suggests the potential development of QoI resistance in South Korea by a mutation in the target gene, cytochrome b (Cytb). Different EC50 values of the strains indicated uninvestigated factors affecting fungicide sensitivity in the strains. This is the first report of fungicide sensitivity of S. vesicarium and S. eturmiunum in South Korea, and further analysis for resistance mechanisms is needed.
Pathogenicity tests
Pathogenicity tests of the fungal strains revealed that S. eturmiunum 19-056 and S. vesicarium 19-238 showed the highest disease severity among the strains (Fig. S1). Evaluated DSI percentage of both strains were 60.0(±8.3) and 59.5(±4.4), respectively. These values were significantly higher than the percentage of other strains (19-130, 19-230, 19-241, KACC44528, and KACC44530) (Fig. 1). Pathogenicity of S. vesicarium on Allium spp. is known as robust and widespread (Basallote‐Ureba et al., 1999; Hay et al., 2019; Anupriya et al., 2021). S. eturmiunum has recently been reported on garlic in South Korea (Dumin et al., 2022). A previous study showed strong postharvest pathogenicity of S. eturmiunum 19-056 on garlic cloves and the same strain showed strong virulence on garlic plants, which makes S. eturmiunum another notable pathogen for garlic (Back et al., 2022).
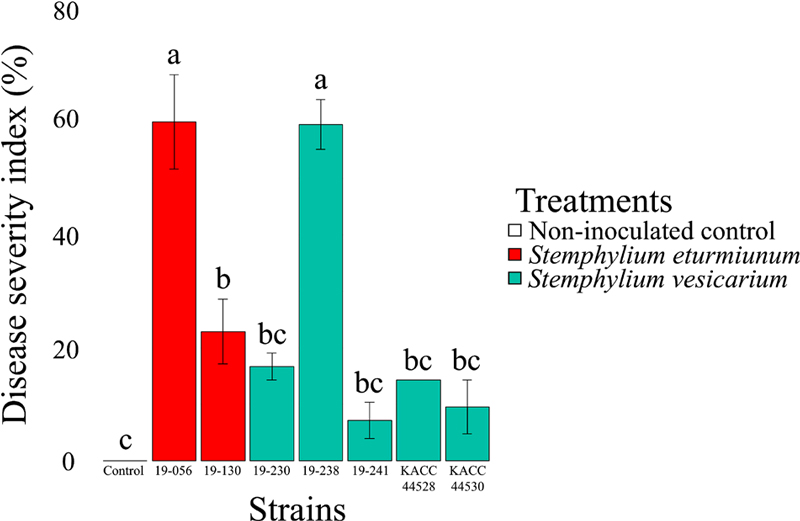
Pathogenicity test of fungal strains. Pathogenicity testing revealed high virulence of some strains in this study. Pathogenicity of 7 strains was tested on garlic plants. Stemphylium eturmiunum 19-056 and Stemphylium vesicarium 19-238 showed significantly higher virulence than other strains with about 60% of the disease severity index (DSI). Virulence of Stemphylium eturmiunum 19-130 was intermediate, and other strains showed no statistically different DSI percentage values.
Fungicide efficacy assays
Evaluation of fungicide efficacy in garlic showed better disease control activity of iprodione compared to the other 4 fungicides. Control efficacy of azoxystrobin, difenoconazole, florylpicoxamid, iprodione, and penthiopyrad against to S. eturmiunum 19-056 were 39.53(±3.89), 27.52(±8.81), 55.23(±6.40), 65.89(±6.55), and 44.57(±9.36), respectively, and to S. vesicarium 19-238 were 10.00(±6.55), 52.73(±7.27), 33.33(±8.99), 82.22(7.03), and 6.67(±11.37), respectively (Fig. 2). To investigate the possibility of using less fungicide, the most effective fungicide iprodione was selected for further experimentation. The average control efficacy of 1X, 2/3X, and 1/2X solutions against S. eturmiunum 19-056 after the 3rd spraying was 46.43(±12.71), 42.86(±10.51), and 39.29(±12.02), respectively, and to S. vesicarium 19-238 were 41.43(±13.53), 42.86(±15.84), and 28.57(±15.03), respectively, (Fig. 3A). Consistency in the amount of sprayed fungicide solutions was confirmed by analyzing surface coverage of water sensitive papers (Fig. 3B). The average percent coverage of water sensitive papers of each spraying of 1X, 2/3X and 1/2X of iprodione was 30.73(±3.69), 31.54(±3.69), and 26.77(±2.68), respectively, which was not statistically different (Fig. 3C).
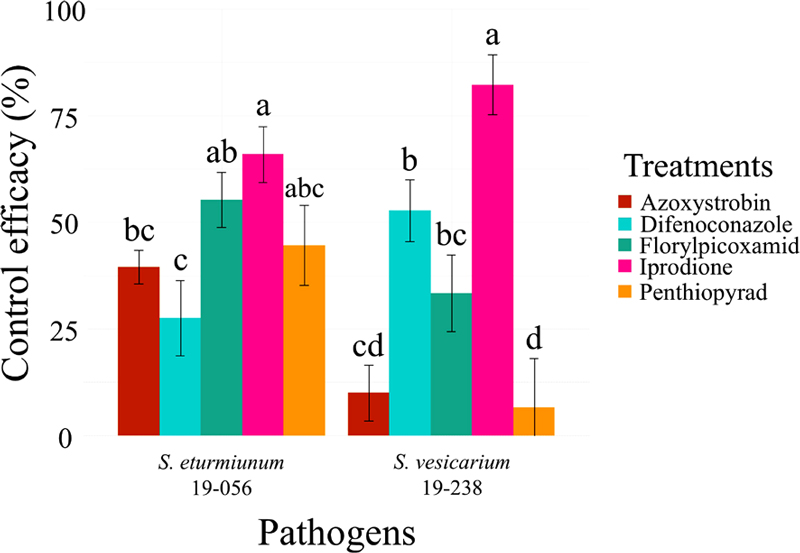
Fungicide efficacy test of 5 fungicides against two pathogens. Azoxystrobin, difenoconazole, florylpicoxamid, iprodione, and penthiopyrad were tested by spraying fungicide solutions at concentrations authorized for UAV application with MSE on garlic (n = 6). Iprodione showed highest control efficacy among the fungicides, while the other fungicides showed different control efficacy depending on the treated pathogen strain. Different letters above the bars indicate statistically significant difference of means determined by Fisher’s LSD test.
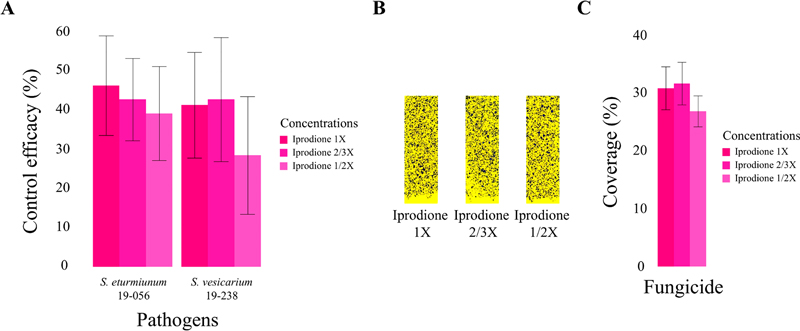
Control efficacy test of diluted iprodione solutions to garlic leaf blight. (A) Control efficacy of iprodione solutions at concentration for UAV spraying (1X) and diluted concentrations (2/3X, 1/2X) was determined. Overall efficacy of diluted solutions was decreased by dilution but was not statistically different. (B) Surface coverage on water sensitive papers was measured to verify consistency in the sprayed amounts of fungicides. (C) Calculated surface coverage on water sensitive paper was not significantly different. Statistically significant differences of means were determined by Fisher’s LSD test.
Each fungicide showed different control efficacy of the treated pathogen strains. Azoxystrobin showed higher control efficacy of 19-056 compared to 19-238, which corresponds to the EC50 value estimation result. In particular, difenoconazole and florylpicoxamid showed high control efficacy despite high EC50 values, indicating the difference between in vitro and in vivo activity of these fungicides. The highest control efficacy of iprodione suggested stable activity of iprodione both in vitro and in vivo. Given the results in this and previous studies, iprodione can be considered as a more reliable fungicide than azoxystrobin against Stemphylium leaf blight (Hay et al., 2019; Spawton et Du Toit, 2024). Reduced concentrations of iprodione – up to half the 1X concentration – were also effective against the pathogens. The results demonstrated that certain fungicides can be sprayed at lower concentrations with equivalent control efficacy and reduced unwanted dispersion of fungicide. Our study can contribute to development of a more efficient and regulated UAV-mediated fungicide spraying system.
Acknowledgments
This research was supported by the Rural Development Administration (grant no. RS-2022-RD010344), Republic of Korea.
Conflicts of interest
The authors declare no conflict of interest.
References
- Anupriya P, Godara SL, Kumhar DR, Chawla N. 2021. Prevalence and management of major diseases of garlic is it global level: A review. Agric. Rev. 43(3):312–319.
-
Back C-G, Han Y-K, Dumin Bin W, Park J-H, Han J-W, Bae Y-S. 2022. Phylogenetic analysis and biological characterization of Stemphylium species isolated from Allium crops. Korean J. Agric. Sci. 49(1):113–120.
[https://doi.org/10.7744/kjoas.20220008]

-
Basallote‐Ureba MJ, Prados‐Ligero AM, Melero‐Vara JM. 1999. Aetiology of leaf spot of garlic and onion caused by Stemphylium vesicarium in Spain. Plant Pathol. 48(1):139–145.
[https://doi.org/10.1046/j.1365-3059.1999.00313.x]

-
Canicattì M, Vallone M. 2024. Drones in vegetable crops: A systematic literature review. Smart Agricultural Technology. 7:100396.
[https://doi.org/10.1016/j.atech.2024.100396]

- Chavda HC, Brahbhatt AB. 2016. Evaluation of fungicides against Aspergillus niger, an incitant of black mold rot of garlic. J. Pl. Dis. Sci. 11(2):217–213.
-
Dumin W, Park M-J, Han Y-K, Bae Y-S, Park J-H, Back C-G. 2022. First report of leaf spot disease caused by Stemphylium eturmiunum on Garlic in Korea. Plant Dis. 106(1):318.
[https://doi.org/10.1094/PDIS-03-21-0674-PDN]

-
Galvez L, Gil-Serna J, Garcia M, Iglesias C, Palmero D. 2016. Stemphylium leaf blight of garlic (Allium sativum) in Spain: Taxonomy and in vitro fungicide response. The Plant Pathol. J. 32(5):388–395.
[https://doi.org/10.5423/PPJ.OA.03.2016.0063]

-
Hay F, Stricker S, Gossen BD, McDonald MR, Heck D, Hoepting C, Sharma S, Pethybridge S. 2021. Stemphylium leaf blight: A re-emerging threat to onion production in eastern North America. Plant Dis. 105(12):3780–3794.
[https://doi.org/10.1094/PDIS-05-21-0903-FE]

-
Koike ST, Smith RF, Davis RM, Nunez JJ, Voss RE. 2011. Characterization and control of garlic rust in California. Plant Dis. 85:585–591.
[https://doi.org/10.1094/PDIS.2001.85.6.585]

- Korean National Statistical Office (KOSTAT). 2023. Production of barley, garlic and onions in 2023. Daejeon, South Korea. p. 5.
-
Mondani L, Chiusa G, Battilani P. Fungi associated with garlic during the cropping season, with focus on Fusarium proliferatum and F. oxysporum. Plant Health Prog. 22:37–46.
[https://doi.org/10.1094/PHP-06-20-0054-RS]

- Park J, Hong S-W, Lee I. 2020. Evaluation of efficiency of livestock vehicle disinfection systems using water-sensitive paper. J. Korean Soc. Agric. Eng. 62(4):87–97.
- R core team. 2021. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/, .
-
Spawton KA, Du Toit LJ. 2024. Prevalence of FRAC group 11 fungicide resistance in Stemphylium vesicarium isolates, but not S. beticola isolates, causing Stemphylium leaf spot of spinach (Spinacia oleracea). Plant Dis. 108(7):2122–2135.
[https://doi.org/10.1094/PDIS-11-23-2328-RE]

- Tattelman EM. 2005. Health Effects of Garlic. Am. Fam. Physician. 72(1):103–106.
-
Valdez JG, Makuch MA, Ordovini AF, Masuelli RW, Overy DP, Piccolo RJ. 2006. First report of Penicillium allii as a field pathogen of garlic (Allium sativum). Plant Pathol.55:583.
[https://doi.org/10.1111/j.1365-3059.2006.01411.x]

-
Wang C-H, Tsai Y-C, Tsai I, Chung C-L, Lin Y-C, Hung T-H, Suwannarach N, Cheewangkoon R, Elgorban AM, Ariyawansa HA. 2021. Stemphylium leaf blight of welsh onion (Allium fistulosum): An emerging disease in Sanxing, Taiwan. Plant Dis. 105(12):4121–4131.
[https://doi.org/10.1094/PDIS-11-20-2329-RE]

Appendix
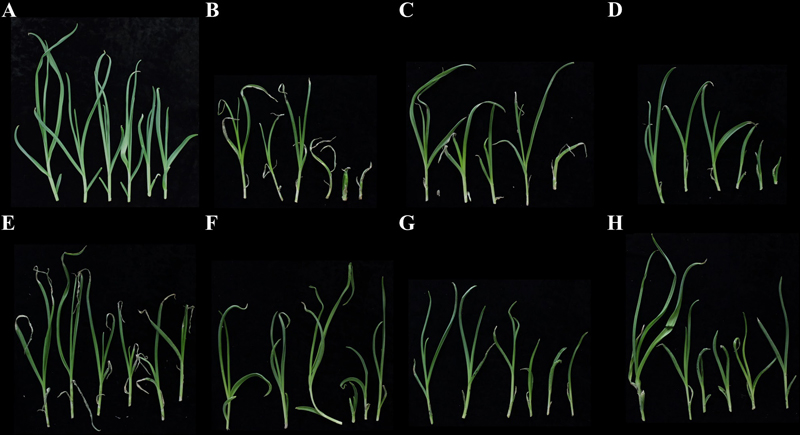
Lesion development in pathogenicity test of Stemphylium eturmiunum and Stemphylium vesicarium strains. Developed lesion on garlic plants by garlic leaf blight strains were shown. Garlic plants were inoculated with no pathogen (A), S. eturmiunum 19-056 and 19-130 (B and C), and S. vesicarium 19-230, 19-238, 19-241, KACC44528, and KACC44530 (D-H), respectively.
Sung-Won Park, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, https://orcid.org/0000-0001-6017-9733
Wooman An, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University
Sungyu Choi, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, https://orcid.org/0000-0003-4447-2491
Hyeongju Choi, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, https://orcid.org/0000-0003-0435-8483
Jae-Ha Choi, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, https://orcid.org/0009-0008-1622-4013
Soyoon Park, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, https://orcid.org/0009-0004-2775-108X
Hyeong-Rok Jang, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, https://orcid.org/0009-0002-6854-6301
Chang-Gi Back, Department of Environmental Horticulture and Landscape Architecture, Environmental Horticulture, Dankook University
Se-Woon Hong, Department of Rural and Bio-Systems Engineering, Chonnam National University, https://orcid.org/0000-0002-9647-5523
Hyunkyu Sang, Department of Integrative Food, Bioscience and Biotechnology, Chonnam National University, https://orcid.org/0000-0002-7459-5217
Experimentation: Sung-won Park, Wooman An, Sungyu Choi, Hyeongju Choi, Jae-Ha Choi, Soyoon Park, Hyeong-Rok Jang Methodology & Writing original draft preparation : Sung-won Park, Wooman An, and Hyunkyu Sang
Data Analysis: Sung-won Park, Wooman An, Chang-Gi Back, and Se-Woon Hong
Writing Review & Editing: Hyunkyu Sang, Chang-Gi Back, and Se-Woon Hong
Conceptualization & Supervising : Hyunkyu Sang
